Drug and diagnostic development both follow rigorous scientific and regulatory journey in the name of clinical quality assurance and adversity mitigation. Current advancements of nanoscopic proteomics platforms are transforming these development cycles by enabling unprecedented precision in target discovery, biomarker validation, and mechanism-of-action studies. The high resolution and precision qualities of nanoscopic proteomics platforms allowed diagnostic researchers to more accurately pinpoint a biomarker of high pathological relevance which was difficult with traditional technologies. This in turn, benefited diagnostics development by enabling higher accuracy and fast-tracked biomarker selection qualification as candidates for follow-up clinical and analytical assessment.
Scientific Journey of Diagnostic Marker Development
- Biomarker Discovery and Validation
- Identification of disease-associated proteins or molecular signatures.
- Nanoscopic proteomics enables:
- Discovery at subcellular resolution.
- High-throughput biomarker screening.
- Integration of proteomics and spatial context to accurately pinpoint subcellular protein interactions and disease biology.
- Clinical Test Development
- Development of assay procedures and technical methods based on biomarkers qualified from the discovery phase.
- High assay reproducibility is a desirable quality
- Test quality will depend on biomarkers qualified from discovery phase
- Analytical Assessment and Optimization
- At this point, the test should possess:
- Good reproducibility
- High sensitivity
- High specificity
- Predictability
- Linear response over the relevant range of measures
- If the test does not check for above qualities, the development cycle must start-over due to poor biomarker selection/qualification from the discovery phase
- At this point, the test should possess:
Role of Nanoscopic Proteomics
Velgen Technik recognizes the critical importance of disease biomarker accuracy which either make or break the development cycle of diagnostics. We offer state-of-the-art nanoscopic proteomics platforms of to accurately capture biomarkers of high biological relevance, offering researchers high confidence in qualifying biomarkers for further diagnostics development while mitigating risks of re-development due to poor biomarker significance.
Velgen Technik Nanoscopic Proteomics Platforms
- Syncell Platform
- Provides label-free, spatial proteomics at nanoscopic resolution.
- Addresses limitations of hypothesis-driven approaches that restrict analysis to known targets.
- Complete coverage of protein identification at subcellular levels such as organelle contact sites, disease-associated microenvironments or subcellular regions that are difficult to isolate
- Accelerates biomarker discovery and de-risks biomarker and drug pipelines by revealing mechanisms of action and off-target effects.
- Hawk Biosystems
- Spatial proteomics with functional context
- Enables mapping of protein interactions in subcellular microenvironments
- Supports precision medicine by linking proteomic signatures to therapeutic response.
Case Studies
- Syncell Platform
The primary cilium is a critical organelle involved in signal transduction, development, and disease. However, its small size, fragile nature, and the limited abundance of associated proteins make comprehensive proteomic profiling challenging. Leveraging the advanced capabilities of the Syncell Microscoop®, to rapidly capture proteins at specified subcellular regions of interest with nanoscale precision. This platform’s high sensitivity and speed allow for the identification and quantification of low-abundance thousands of proteins with subcellular resolution providing new insights into subcellular protein dynamics and functions.
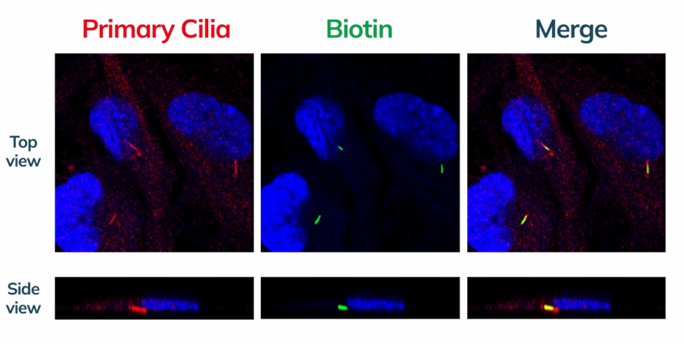
Primary cilia (red) recognized as region of interest via fluorescent staining for subsequent photolabeling (left panel, also control). Photolabeled primary cilia (green, middle panel), expected to overlap with red (right panel, demonstrates accurate photolabelling). Red: GT335, Green: NeutrAvidin-488, Blue: DAPI. Proteins within the photolabelled region (primary cilia) is subsequently pulled-down for identification.
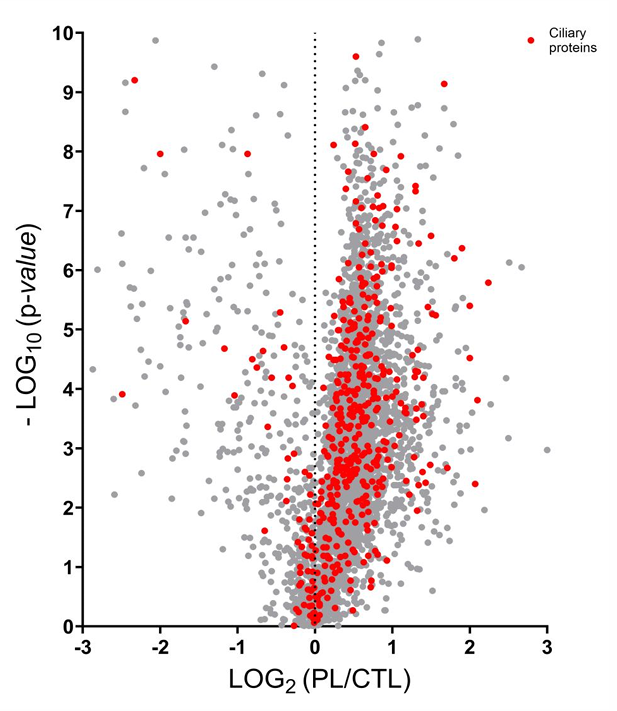
Proteins identified from primary cilia are as depicted as red dots above. Distribution of overall protein abundances is binned by the ratio of copies in a photolabeled (PL) sample to those in a control (CTL) sample annotated as PL/CTL ratio. Ciliary proteins (red) are enriched in the PL group compared to the CTL sample.
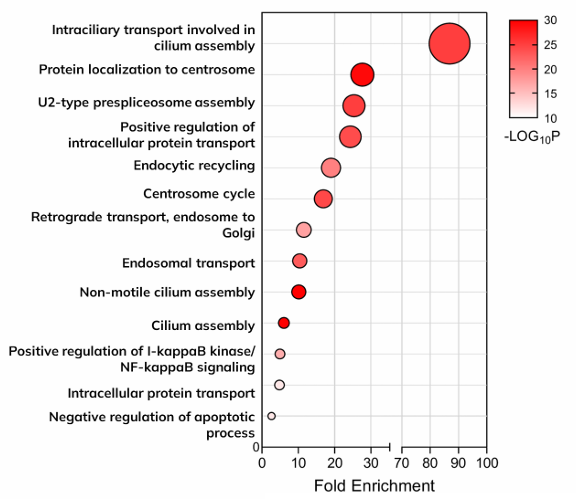
The top 100 enriched proteins were subjected to Gene ontology to reveal cilia related biological process.
427 enriched ciliary proteins were subjected to Reactome to reveal cilia related pathways to contextualize cilial proteins with and their relevance to disease mechanisms.
- Hawk Biosystems
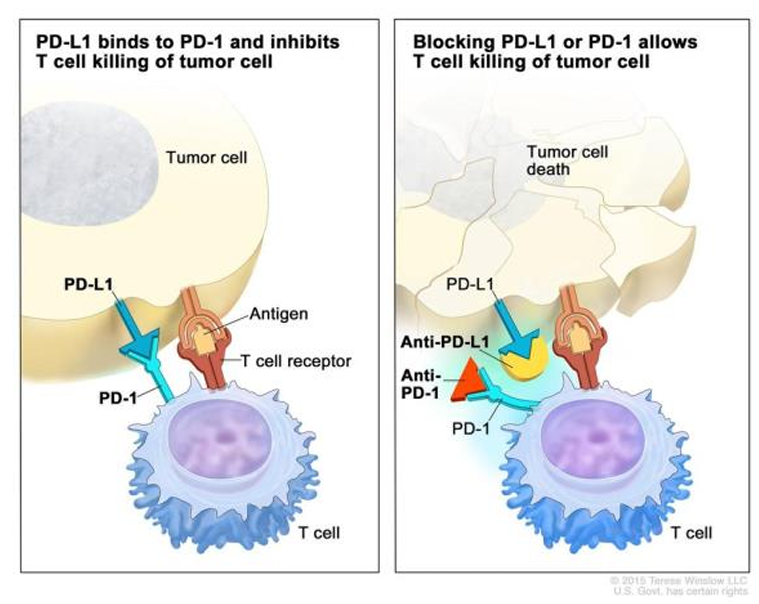
PD-1/PD-L1 is an immune checkpoint designed to switch off the immune system to prevent auto-immune disease. However, some cancers upregulate the ligand to evade the immune system, which is inherently programmed to destroy neoplastic cells. Drugs blocking these interactions between the immune system and cancer cells can allow the immune system to find and clear cancer cells from the body.
Currently, non-small cell lung cancer (NSCLC) patients are selected for ICI therapy based on the expression levels of the checkpoint ligand. Within our case study, we analysed arguably the most well-known immune checkpoint, PD-1/PD-L1. The expression of the ligand, PD-L1 is used to select patients for ICI therapies aimed at blocking PD-1/PD-L1 interactions. However, this approach has several limitations which resulted in two groups of mistreated patients. One group of patients presented with high ligand expression but do not have significant checkpoint interaction, therefore are receiving treatments that will not benefit them (and are missing out on treatments that would). The second group of patients present with lower PD-L1 levels, but, despite this, the PD-L1 present in their samples is interacting significantly with the receptor PD-1. These patients are excluded from ICI therapies that would greatly benefit them.
In contrast to quantifying expression, Hawk Biosystems’ QF-Pro® assay quantifies PD-1/PD-L1 interaction instead which is the more accurate biomarker that is representative of the biological mechanisms of the immune checkpoint.
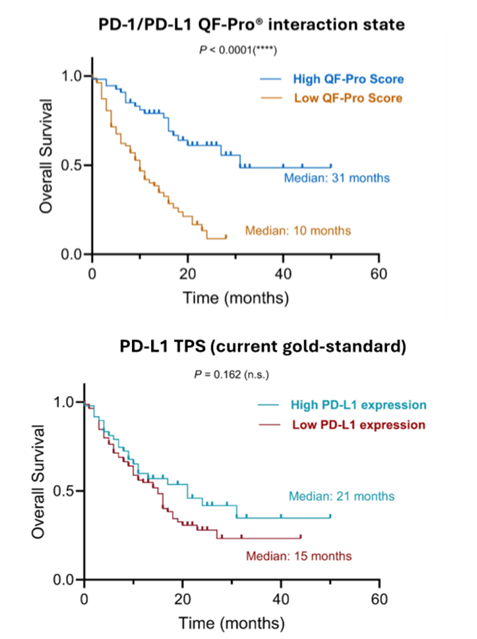
High PD-1/PD-L1 interaction state, quantified with QF-Pro® stratified patients into high and low interaction states which correlated significantly with overall survival in response to immunotherapy, compared to the currently gold-standard via PD-L1 expression. This study signified the importance of biomarker accuracy within diagnostics that will impact treatment decisions.
Sources:
- Deep Spatial Proteomics of Primary Cilia: Unlocking Subcellular Proteome Composition with Nanoscale Precision [https://www.syncell.com/wp-content/uploads/Deep-Spatial-Proteomics-of-Primary-Cilia-Unlocking-Subcellular-Proteome-Composition-with-Nanoscale-Precision.pdf]
- Immune Checkpoint Interaction Quantification by QF-Pro® Whitepaper [https://www.hawkbiosystems.com/whitepapers/immune-checkpoint-interaction-quantification-by-qf-pro-whitepaper/]
- Commercializing Successful Biomedical Technologies: Basic Principles for the Development of Drugs, Diagnostics, and Devices


